 
Element/Moleculeīy knowing the molar mass of a substance, having any mass of that substance allows one to know how many moles of it they have. Molar mass of CO is 28.0101 g/mol Compound name is carbon monoxide Get control of 2022 Track your food intake, exercise, sleep and meditation for free. A list of some molar masses can be seen below. Get control of 2022 Track your food intake, exercise, sleep and meditation for free.  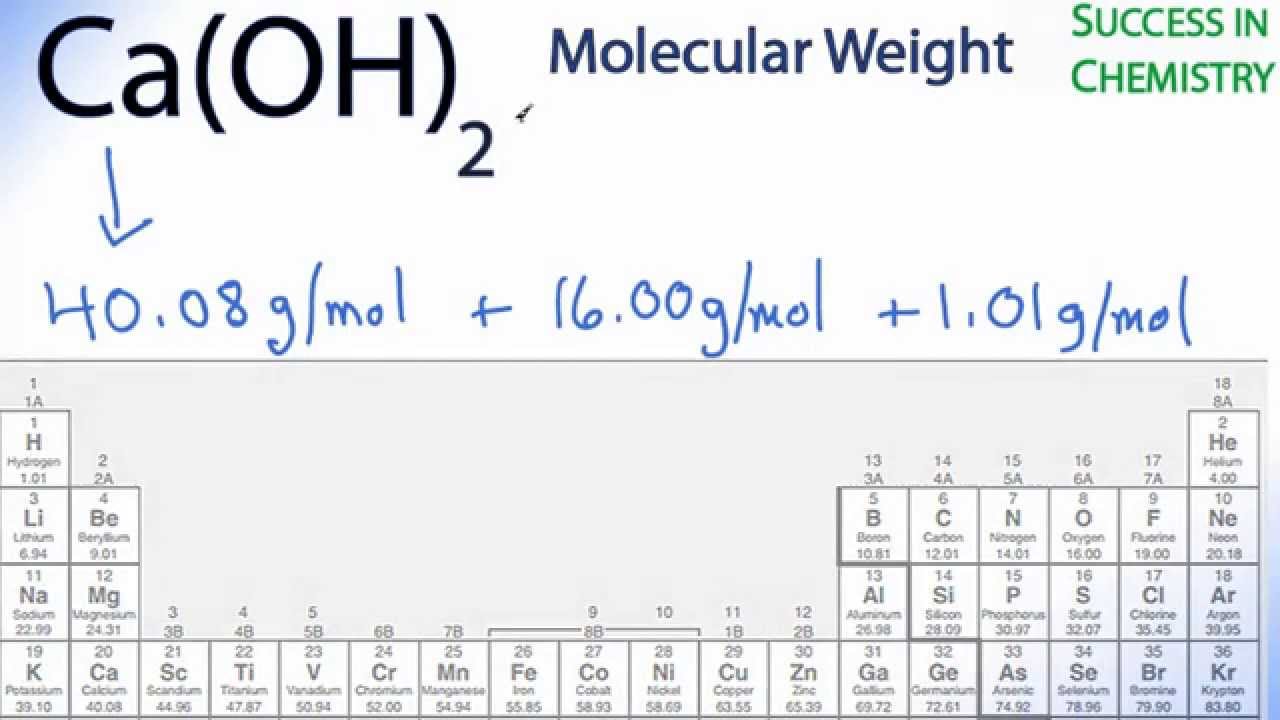
The molar mass of any element or molecule is given by the sum of the atomic weights multiplied by 1 g/mol. Molar Mass, Molecular Weight and Elemental Composition Calculator. In SI units, the molar mass is given by kg/ mol, however by convention the molar mass is expressed in units of g/mol. This comparison is seen below:ġ dozen = 12 objects 1 mol = 6.022141 x 10 23 objects However when talking about moles, instead of only having a dozen, there is in fact 6.022141 x 10 23 of a given substance in a mole (known as Avogadro's number). Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. For example, if someone has a dozen bricks and another person has a dozen feathers, they have the same amount of their substance, but a vastly different total mass. Element Cobalt (Co), Group 9, Atomic Number 27, d-block, Mass 58.933. Since a mole is defined as the amount of a substance and substances have different masses, each element or molecule will have a different molar mass. Molar mass (M) is a physical property, defined as the mass of a given element or molecule per mole of that substance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
